
Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) are one of the most common medications used by many to treat mild to moderate pain. As some of them are considered over-the-counter drugs, they’ve become increasingly relied upon by the average consumer for quick pain relief. As such, understanding how NSAIDs work and what sets them apart from other medications can be extremely valuable for students interested in pharmacology.
During your pharmaceutical or regulatory affairs program, you’ll be introduced to key regulatory systems, legislations, and practices that directly affect the development and quality of pharmaceutical products. Here’s a closer look at NSAIDs for those studying pharmaceutical regulatory affairs.

NSAIDs are one of the most common medications for quick pain relief
What NSAIDs Are Used for and How They Work
NSAIDs make up a large group of drugs that consist of various products developed with different chemical structures. Despite the range of NSAIDs—with some needing a prescription and others being sold over the counter—they all work to reduce pain, inflammation, and high fever. They can be used to treat a variety of discomforts, including:
- headaches
- swelling
- muscle or joint pain
- sprains
- menstrual cramps
- toothaches
- cold or flu symptoms
As their name suggests, NSAIDs act as anti-inflammatory drugs by blocking the enzymes (Cox-1 and Cox-2) that help create prostaglandins, a chemical compound that’s directly linked to the body’s inflammatory response process. By slowing the production of this chemical, NSAIDs can provide quick pain relief.

NSAIDs can help relieve mild to moderate pain from common discomforts such as headaches
NSAID Side Effects and Precautions That Students in Pharmaceutical Training Should Know About
Students taking pharmaceutical courses will do well to note that studying side effects and possible allergic reactions to medications is important. NSAIDs are great pain relievers, but prolonged usage can lead to a number of side effects, varying from mild to more severe conditions. These include:
- dizziness or drowsiness
- nausea or vomiting
- allergic reactions (linked to asthma) or shortness of breath
- gastrointestinal problems
- high blood pressure
- fluid retention (edema)
- kidney damage (reducing blood flow)
As NSAIDs serve to counteract the development of prostaglandins (which are important for helping platelets with blood clotting and protecting the stomach and GI lining from damage), the use of these medications can result in additional side effects. For instance, chronic use of NSAIDs might lead to ulcers or prolonged bleeding after injuries or surgeries. This side effect makes many NSAIDs (apart from low-dose aspirin) potentially harmful for people with heart conditions or blood vessel diseases.
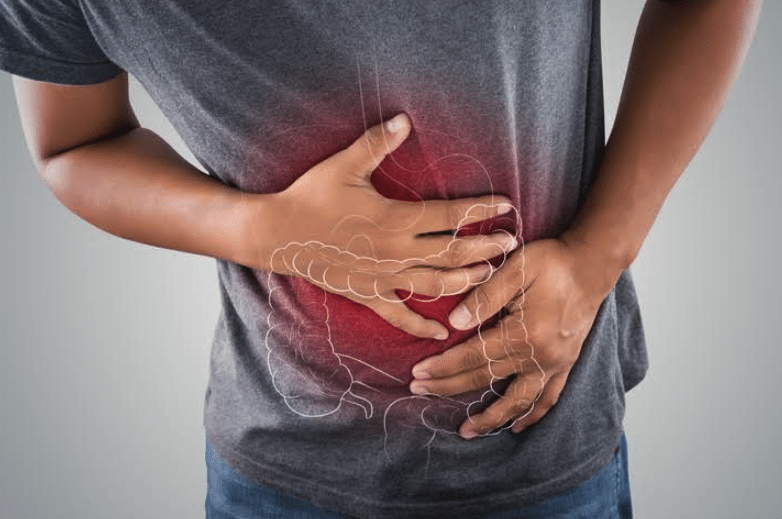
Prolonged use of NSAIDs can lead to some gastrointestinal problems
Understanding the Difference Between Various NSAIDs
The availability of various NSAIDs makes it increasingly important to understand the differences between them. Prescription NSAIDs are typically more powerful than over-the-counter (OTC) versions, with commonly prescribed examples including Voltaren (Diclofenac) and Indomethacin. OTC NSAIDs include ibuprofen and aspirin. Unlike other NSAIDs, aspirin has the additional effect of counteracting blood clotting—making it particularly useful for preventing strokes and heart attacks.
Pharmaceutical regulatory affairs professionals will understand that not all NSAIDs work in the same way. Some affect both Cox-1 and Cox-2 enzymes, while others serve only as Cox-2 inhibitors. These NSAIDs relieve pain similarly, but Cox-2 inhibitors (such as Celebrex) were specifically engineered to avoid gastrointestinal side effects. Blocking either enzyme can lead to some imbalances, since they’re naturally regulated together. Knowing these effects and the detailed differences between various pharmaceutical products is an important part of work in the industry.
Are you interested in starting your pharmaceutical training?
Contact AAPS for more information!
